 study the information in the table below and answer the questions below the tableĬalculate the enthalpy change of the reaction: Calculate the relative atomic mass of element J given that the specific heat capacity of water=#4.2KJ^(-1)g^-1#, density of water=#1.0gcm^-3# and molar 
When 0.6g of element J were completely burnt in oxygen and all the heat evolved was used to heat #500cm^3# of water, the temperature of the water rose from #23^0C# to #32^0C#. (b) Write an expression, for #DeltaH_3# in terms of #DeltaH_1# and #DeltaH_2# Study the diagram and answer the questions that follow. A simple energy level diagram for the reaction is given below. Sulphur burns in air to form sulphur (IV) oxide. (b) Calculate the molar enthalpy of combustion, #DeltaH4# of ethanol (a) Define the term “enthalpy of formation” of a compound. Use the information below to answer the questions that follow: In an experiment determine the heat of combustion of methanol, #CH_3OH#, a student used a set up like the one shown in the diagram below. 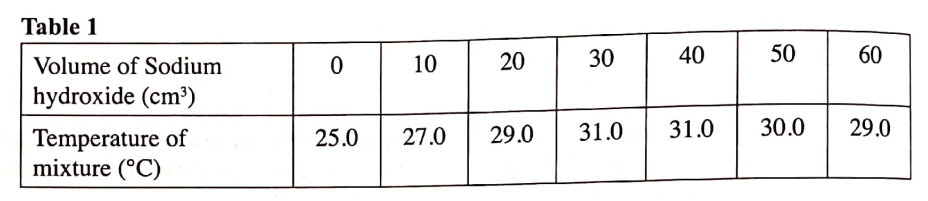  
Explain why the enthalpy of neutralization of ethanoic acid with sodium hydroxide is different from that of hydrochloric acid with sodium hydroxide. (b) What does the sign #DeltaHc# value indicate about combustion of alkanes? (a) Predict the heat of combustion of butane and write it on the space provided in the table above. Study the table below and answer the questions that follow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
